When Cancer Changes Identity
A new study co-led by ISB and published in the journal Cell Reports Medicine reveals how a rare lung cancer can shift between cell types, pass through hybrid states, and build distinct tumor “neighborhoods” that may help it evade detection and resist treatment.
Most cancers are defined by what they are. But some are defined by what they can become.
In a new study published in Cell Reports Medicine, researchers show that a rare form of lung cancer can change identity as it grows — shifting from one cancer type to another and passing through unstable, hybrid states along the way. The work suggests that this cancer is not a static mass, but a moving target.
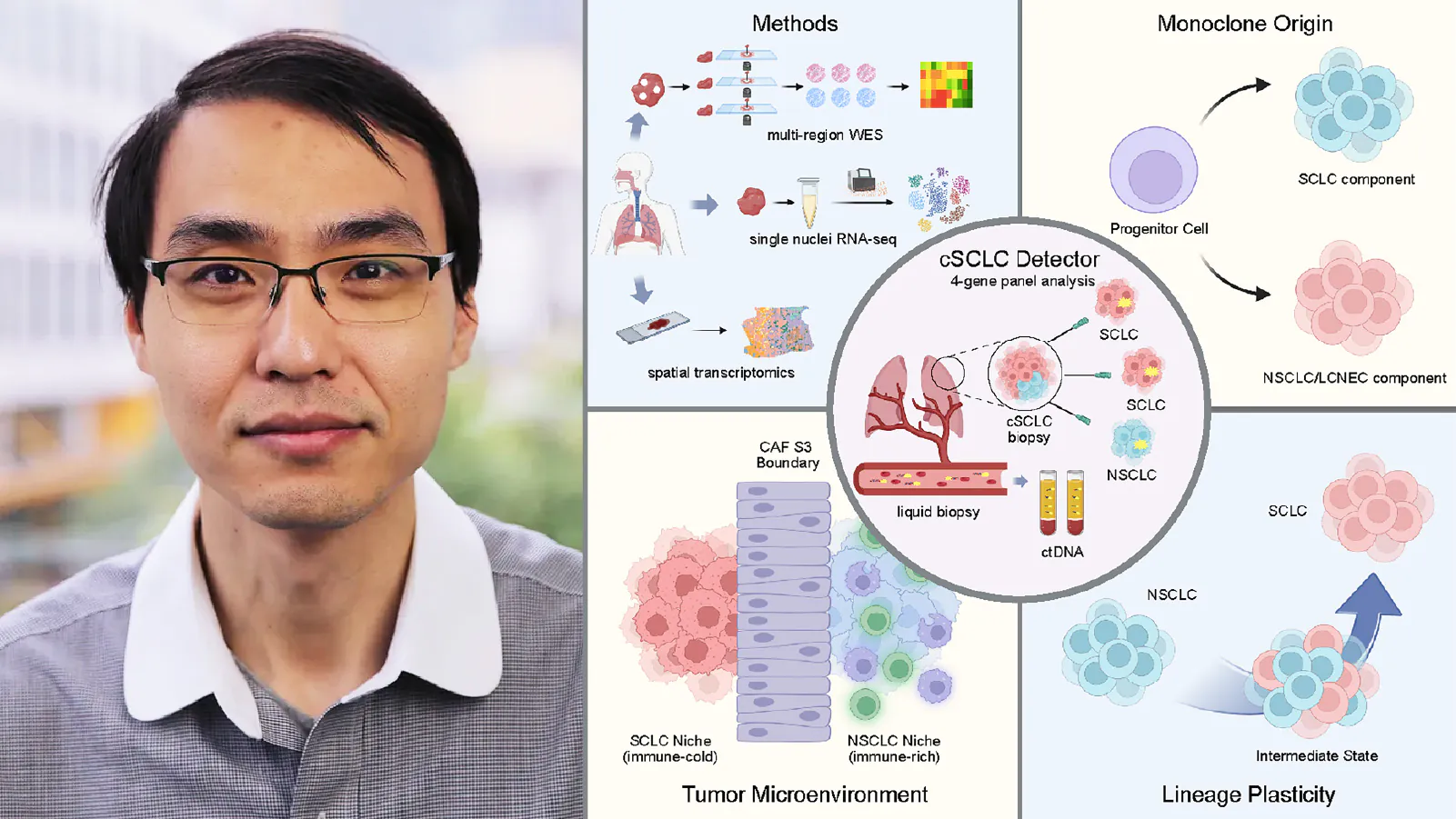
The work focuses on combined small-cell lung cancer (cSCLC), a rare and aggressive disease in which tumors contain features of both small-cell and non-small-cell lung cancer. Because these mixed tumors are difficult to detect in small biopsies, they are often treated as standard small-cell lung cancer, even though patients tend to have worse outcomes. That mismatch matters because a tumor that looks like one disease under the microscope may in fact be something more complex and potentially more dangerous.
The key question: Are these tumors a collision of two separate cancers that happen to grow side by side — or a single cancer evolving into different forms?
The answer, the researchers found, is the latter.
Using a combination of spatial genomics, single-cell analysis, and multi-region sequencing, the team showed that these tumors arise from a single ancestral cancer cell. The different tumor regions may look very different, but they trace back to the same origin and then branch into distinct identities.
“We found that these tumors are not simply mixtures of different cancer types,” said Wei Wei, PhD, associate professor at ISB and co-corresponding author of the study. “They are dynamic systems, with cancer cells actively changing their identity.”
A tumor in transition
The study provides one of the clearest pictures to date of what researchers call lineage plasticity — the ability of cancer cells to change identity.
Rather than switching cleanly from one type to another, tumor cells were found along a continuum. Some cells retained features of both small-cell and non-small-cell lung cancer at the same time. In other words, the change was not a simple flip of a switch. It looked more like cells moving through unstable in-between states on the way from one identity to another. About one-third of the SCLC-like tumor cells analyzed fell into these intermediate, hybrid states.
That finding suggests cancer progression is not a binary process, but a gradual transition — one that may allow tumors to adapt, evade treatment, and become more aggressive.
“If cancer cells can shift from one identity to another, then therapies aimed at today’s tumor may miss the version it becomes tomorrow,” Wei said.
A tumor made of neighborhoods
The study also reveals that these tumors are not uniform. Instead, they are organized into distinct regions, each with its own biological environment.
Some areas — particularly those resembling non-small-cell lung cancer — were rich in immune cells. Others, especially small-cell-like regions, were largely immune-excluded.
These regions were often separated by dense bands of fibroblasts, structural cells that produce collagen and help shape tissue architecture. In these tumors, fibroblast-rich boundaries may act as physical and biochemical barriers, limiting immune access to certain regions.
The result is a tumor composed of “hot” and “cold” neighborhoods — areas where immune cells are active, and others where they are effectively shut out.
That patchwork organization may be one reason why the tumor behaves so aggressively and why immunotherapy responses are uneven across the same lesion.
A hidden cancer, hiding in plain sight
The study also points to a practical challenge: many of these tumors may be going undetected.
In cSCLC, the different tumor regions are often physically separated, and the small-cell component frequently dominates the tumor mass. That makes it easy for a biopsy to sample only one region — potentially missing the mixed nature of the disease.
To address this, the researchers developed a four-gene diagnostic tool, called cSCLC Detector, designed to identify these tumors more accurately. The logic came directly from the tumor’s evolutionary history: because the small-cell and non-small-cell parts share a common ancestor, they also share early trunk mutations. That means even a biopsy that captures only the small-cell-looking region can still carry genetic clues that reveal the tumor’s hidden mixed identity.
When applied to existing datasets of patients diagnosed with standard small-cell lung cancer, the tool identified a substantially higher proportion of cases with combined features than traditional pathology estimates suggest.
That raises a provocative possibility: this “rare” cancer may be more common than currently recognized — and simply underdiagnosed.
Beyond mutations: understanding cancer as a system
The findings highlight a broader shift in how scientists think about cancer.
Cancer is often described as a genetic disease — and it is. But this work shows that it is also a disease of cellular identity and environment.
Who a cancer cell is becoming, and where it lives within a tumor, may matter as much as the mutations it carries.
For patients, that could eventually mean more accurate diagnoses, earlier detection of dangerous transitions, and treatments designed not only to target the cancer that is visible today, but to anticipate what it may become next.
The collaborative study, “Spatial multi-omics unveils the mono-clonal origin, neuroendocrine plasticity, and microenvironment niches in combined small-cell lung cancer,” was published in Cell Reports Medicine.


