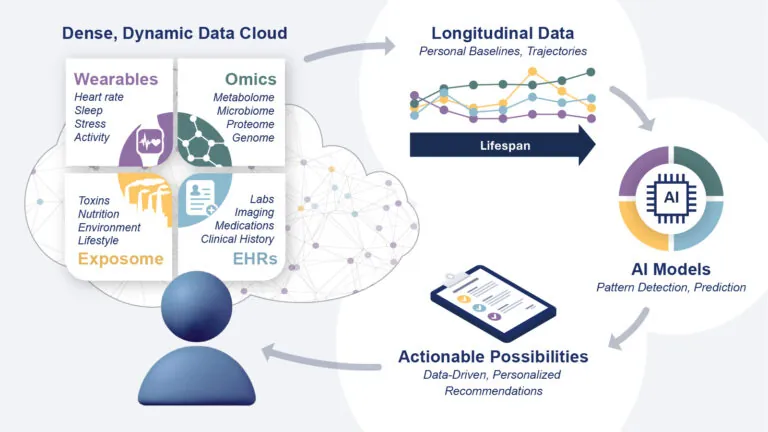
Understanding what it means to be healthy is the key to slowing disease.
Research suggests that it may be possible to eliminate the correlation between aging and disease. ISB is pioneering a new approach to health by focusing on the biological factors that influence wellness throughout your life. Our goal is to create personalized strategies that will improve health, prevent disease, and enhance quality of life for individuals and communities worldwide.