This website uses cookies so that we can provide you with the best user experience possible. Cookie information is stored in your browser and performs functions such as recognising you when you return to our website and helping our team to understand which sections of the website you find most interesting and useful.
Healthy Aging
Science Transforming Health
 isbscience.org/research/healthy-aging/
isbscience.org/research/healthy-aging/![]()
Understanding aging is the key to slowing all diseases
Research suggests that it may be possible to eliminate the correlation between age and diseases. ISB is working to leverage systems biology approaches to understand the mechanistic links among the processes that accompany and/or lead to aging.
“The biggest risk factor for disease is chronological age, and there are core mechanisms associated with aging that underlie many diseases. If we can understand and target those core aging factors, we can delay the onset of all diseases rather than only one specific disease.”
– Dr. Alice Kane, Ling/Obrzut Assistant Professor, ISB
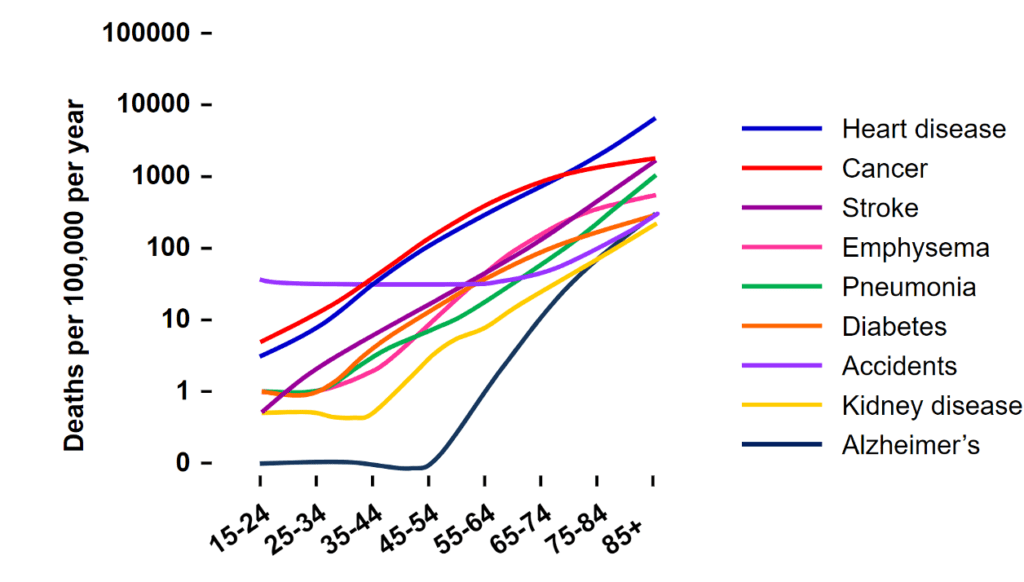
Image Credit: Steve Cummings, 2019
The Hallmarks of Aging
Humans have grown accustomed to the symptoms of aging: increased mortality, decreased fitness, loss of function, damage accumulation, and the general process of becoming old. The incidence rates of heart disease, cancer, Alzheimer’s and nearly every other chronic disease increase sharply as we age. However, researchers have uncovered key regulators that control the aging process, called the “hallmarks of aging”.
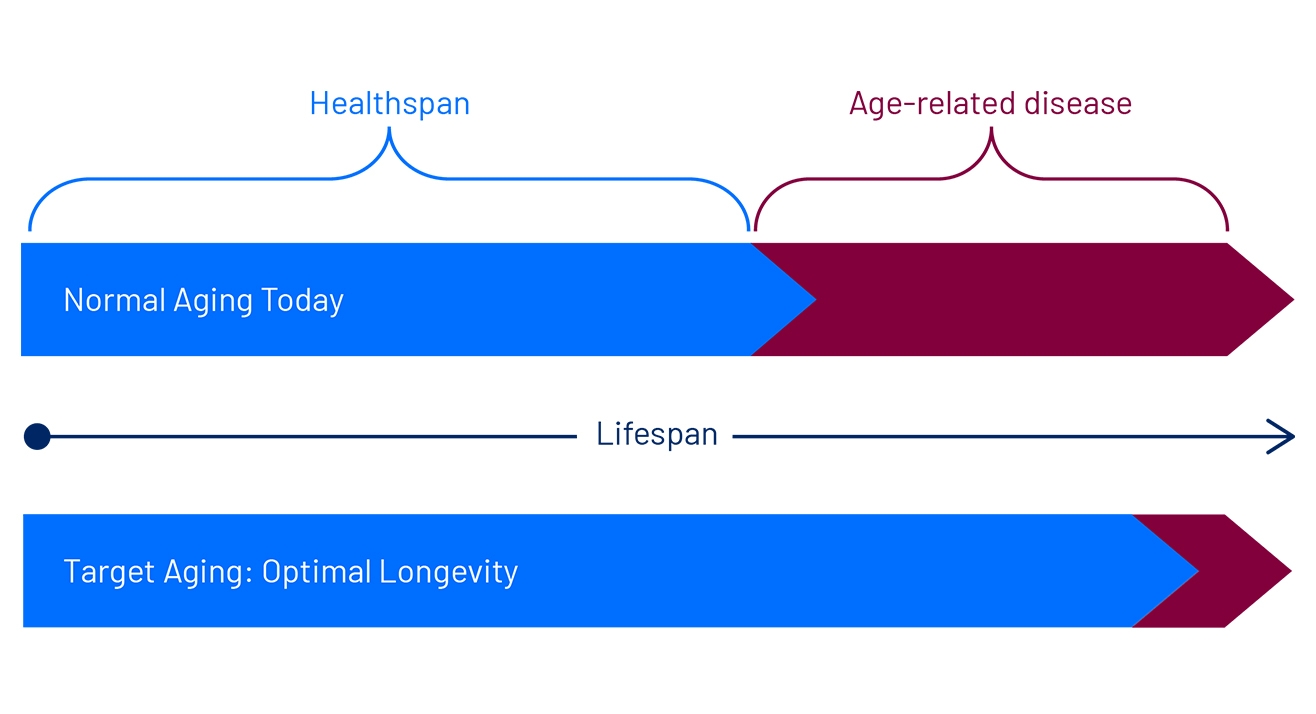
Extending Healthspan to Optimize Longevity
Healthspan is the idea of the period of one’s life within their lifespan where they are in relatively good health. Most people have a period of time at the end of their life where they have age-related diseases, and they have lost independence and function. The aim is to study the mechanisms that factor into aging so that we can maximize our period of healthspan.
A Root Component in Health
ISB researchers have learned to harness deep molecular and physiological information to determine a person’s biological age. Unlike your chronological age (the number of birthdays you’ve celebrated), researchers have found that biological age can serve as an effective and reliable predictor of overall health, and they are getting to the root of aging and determining ways to slow its progression. We do this by working to understand how aging processes, the immune system, the gut microbiome, and other systems affect our ability to modify aging.
Longitudinal Studies
RESEARCH
We are exploring the use of longitudinal multi-omics to translate discoveries from model organisms to human clinical trials and mitigate varied responses to different anti-aging interventions.
Longevity Consortium
RESEARCH
ISB is a core member of the National Institute on Aging’s (NIA) Longevity Consortium. NIA was set up by the National Institutes of Health to lead a broad scientific effort to understand the nature of aging and to extend the healthy, active years of life. NIA is also the primary federal agency supporting and conducting Alzheimer’s disease research.









